
Instructions pour l’écouvillonnage nasopharyngé et la commande de trousses
Conformément au Règlement de l’Ontario 671/92 de la Loi sur les services en français, les formulaires de renseignements d’analyses de laboratoire liés à la présente page ne sont offerts qu’en anglais parce qu’ils sont de nature scientifique ou technique et destinés uniquement à l’usage des fournisseurs de soins de santé qualifiés et non aux membres du public.
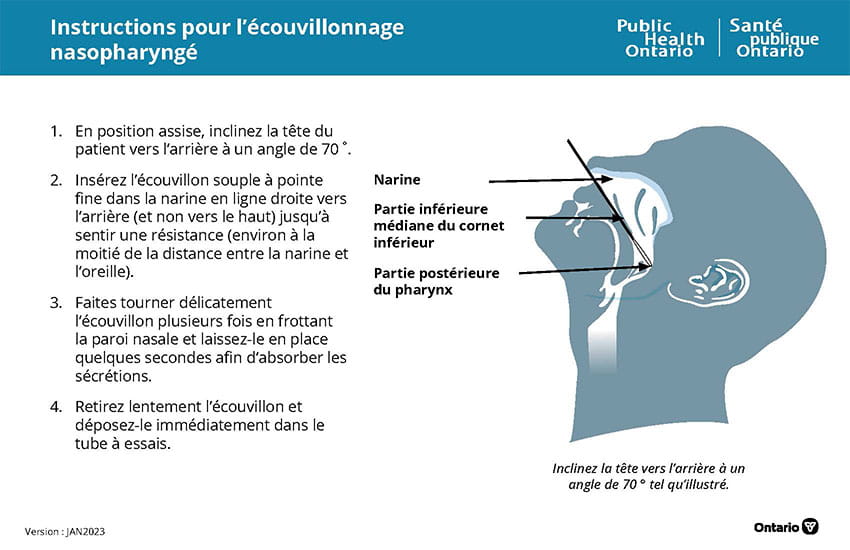
Directives
- Le patient doit ÉVITER de manger, boire, mâcher de la gomme, fumer ou vapoter au moins 30 minutes avant le prélèvement.
- Lavez-vous les mains et portez l’équipement de protection individuelle (EPI) approprié (blouse, gants, masque, visière).
- Inscrivez la date de prélèvement, le nom complet du patient et un autre identifiant unique comme sa date de naissance ou son numéro de carte Santé sur l’étiquette du tube.
- Si vous ne fournissez pas ces renseignements, l’analyse pourrait être refusée ou retardée.
- Remplissez tous les champs du formulaire de demande d’analyse de la COVID-19 et des virus respiratoires.
- Si vous ne fournissez pas ces renseignements, l’analyse pourrait être refusée ou retardée.
- Retirez l’écouvillon de son emballage en vous assurant de ne le tenir que par le manche.
- Recueillez l’échantillon en suivant la procédure illustrée ci-dessus.
- Placez l’embout de l’écouvillon dans le tube.
- Cassez le manche de l’écouvillon de manière uniforme à l’endroit prévu à cet effet.
- Ne pliez pas le manche de l’écouvillon au lieu de le casser.
- Remettez le bouchon du tube en vissant bien serré.
- Si vous ne rebouchez pas le tube correctement, celui-ci pourrait couler et l’analyse pourrait être refusée.
- Placez l’échantillon dans le compartiment principal du sac pour matières contaminées.
- Placez le formulaire de demande d’analyse dûment rempli dans la pochette extérieure du sac pour matières contaminées, pour qu’il ne soit pas exposé à l’échantillon.
- Lavez-vous les mains et envoyez l’échantillon au laboratoire de la manière qui convient.
- Pour une conservation optimale, l’échantillon doit être entreposé et transporté à une température de 2 à 8 °C. Si le transport au laboratoire est retardé au-delà de 72 heures, l’échantillon doit être congelé à -70 °C ou moins et livré sur de la glace sèche.
Entreposage des trousses non utilisées
Les trousses non utilisées doivent être entreposées à une température de 2 à 25 °C jusqu’à leur utilisation. Au-delà de ces températures, elles perdront de leur efficacité. Les trousses ne doivent pas être conservées après leur date d’expiration.
Certaines trousses de prélèvement peuvent disposer de prolongations de leur date d'expiration allant au-delà de l'étiquette du fabricant. La liste des prolongations de la date d'expiration des produits de dépistage de la COVID-19 est disponible sur la page des documents sur l’intervention du système de santé dans la lutte contre la COVID-19.
Limites
Il faut privilégier les écouvillons conçus pour les prélèvements nasopharyngés (pour en savoir plus, voir la page de SPO sur les trousses de prélèvement pour les tests PCR de la COVID‑19). La prudence et le jugement clinique sont de mise lors de l’écouvillonnage chez des personnes ayant des traumatismes faciaux récents, souffrant d’épistaxis sévère ou présentant une anomalie considérable de l’anatomie nasopharyngée.
Autres ressources
- Marty, F. M., K. Chen, K. A. Verrill. « How to obtain a nasopharyngeal swab specimen » [vidéo sur Internet], Waltham (Massachusetts) : The New England Journal of Medicine, 2020 [cité le 1er février 2021]. 5 minutes.
- Centers for Disease Control and Prevention. Nasopharyngeal (NP) specimen collection steps [En ligne]. Atlanta (Géorgie) : Centers for Disease Control and Prevention, 2020 [cité le 1er février 2021].
Vous n'avez pas de compte MonSPO? S'inscrire maintenant

