Hepatitis A – Serology
Conformément au Règlement de l’Ontario 671/92 de la Loi sur les services en français, les renseignements d’analyses de laboratoire liés à la présente page ne sont offerts qu’en anglais parce qu’ils sont de nature scientifique ou technique et destinés uniquement à l’usage des fournisseurs de soins de santé qualifiés et non aux membres du public.
Background
This page provides testing information for hepatitis A virus (HAV) serology at Public Health Ontario (PHO), including HAV IgM and IgG. For information regarding other testing options, refer to the following PHO webpages:
Updates
- Added webpage for Hepatitis A – Genotyping-Subtyping
- Added an interpretation table
- Updated the submission and collection notes to outline ordering instructions for HAV serology
Testing Indications
Testing for hepatitis A virus (HAV) serology may be indicated:
- for the investigation of patients with suspected acute viral hepatitis
- to determine immune status (note: post-vaccine serologic testing is not routinely recommended)
- as part of an epidemiologic investigation (e.g. outbreak investigation).
Acceptance/Rejection Criteria
- Donor testing is not available through PHO’s laboratory. Specimens from patients being screened as potential donors (e.g. organ, tissue, cells, fertility, etc.) should be referred to a laboratory that performs donor screening assays. Specimens received for donor screening at PHO’s laboratory will be rejected.
- Specimens beyond 14 days from date of collection will not be tested.
Specimen Requirements
| Test Requested | Required Requisition(s) | Specimen Type | Minimum Volume | Collection Kit |
Hepatitis A Serology |
Serum |
IgG only: 1 mL IgM only or IgM+IgG: 2 mL |
Serum Separator Tube (SST) |
Submission and Collection Notes
PHO prefers centrifuged SST for serological testing. Generally, only one FULL draw 5 mL SST is needed for testing a combination of up to six infectious disease serology markers. If a full tube cannot be drawn, submit two tubes. If there is a high clinical suspicion that the patient has an HCV infection and testing for other serological markers is needed, consider submitting two SST to ensure there is adequate volume. Refer to Serology Testing – Laboratory Specimen Collection and Submission Instructions for more information.
Do not submit glass tubes.
Label the specimen container(s) with the patient’s first and last name, date of collection and one other unique identifier such as the patient’s date of birth or Health Card Number. For additional information see: Criteria for Acceptance of Patient Specimens. Failure to provide this information may result in rejection or testing delay.
ALL sections of the General Test Requisition must be completed by authorized health care providers for each specimen submitted, or testing may be delayed or cancelled. Please follow instructions for HAV serology test ordering below.
| Requisition selection(s) | Tests Performed | Notes | Requisition Demonstration |
|---|---|---|---|
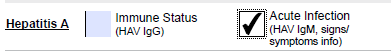
| Immune Status | HAV IgG | 
|
|
| Acute Infection | HAV IgM | Clinical information pertaining to acute viral hepatitis must be provided. If missing, only HAV IgG will be performed. |
|
|
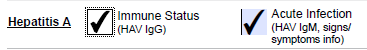
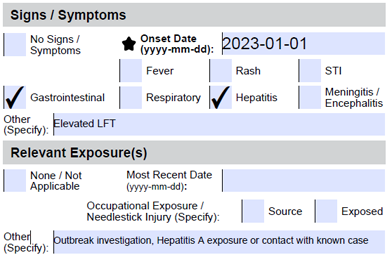
Acute infection and Immune Status |
HAV IgG HAV IgM |
Indicate clinical information pertaining to a recent HAV exposure, outbreak investigation, contact with known case etc. under ‘Relevant Exposure(s)’ Clinical information and/or exposure information (if asymptomatic) must be provided. If missing, only HAV IgG will be performed. |
|
Timing of Specimen Collection
HAV IgM is detectable within 2-4 weeks of infection (5-10 days before the onset of symptoms), peaks within 1-2 months and and may persist for up to 6 months. In rare instances, it can be detected for up to 1 year after an acute infection.
HAV IgG begins to rise at the onset of clinical illness and persists for life.
Limitations
HAV IgM may be detected due to cross-reactivity to non-specific antibody, therefore testing should only be done for individuals with symptoms of acute hepatitis A or who have been exposed to HAV. HAV IgM can also be detected in individuals recently vaccinated with the hepatitis A vaccine.
Haemolysed, icteric, lipemic or microbially contaminated serum is not recommended for testing.
Storage and Transport
Store specimens at 2-8°C following collection and centrifugation and ship to PHO’s laboratory on ice packs within 3 days of collection. If delayed shipping is anticipated, remove serum from clot and store frozen at -20°C or colder and ship on dry ice. Place specimen tube in biohazard bag and seal. Place completed General Test Requisition in the pouch at the front of the biohazard bag. All clinical specimens must be shipped in accordance to the Transportation of Dangerous Good Act.
Test Frequency and Turnaround Time (TAT)
HAV serology testing is performed daily Monday to Friday at PHO’s laboratory. Turnaround time is up to 3 business days from receipt at PHO’s laboratory.
- HAV genotyping is performed at the National Microbiology Laboratory (NML). Refer to Hepatitis A – Genotyping-Subtyping for more information
Specimens for HAV serology are tested using a chemiluminescent microparticle immunoassay (CMIA) for the qualitative detection of IgG and/or IgM antibodies.
Algorithm
Refer to the HAV serology test ordering instructions above for when IgM and IgG are tested.
All positive HAV IgM specimens are automatically sent to the National Microbiology Laboratory for RNA detection and genotyping.
Interpretation
The following table provides possible test results with associated interpretations:
|
Hepatitis A IgG Result |
Hepatitis A IgM Result |
Interpretation |
|---|---|---|
|
Non-reactive |
Not tested |
No evidence of immunity. |
|
Reactive |
Not tested |
Evidence of immunity. |
|
Not tested |
Non-reactive |
No evidence of recent infection/recent vaccination. |
|
Not tested |
Reactive |
Evidence of recent infection or vaccination. |
|
Non-reactive |
Non-reactive |
No evidence of immunity. No evidence of recent infection/recent vaccination |
|
Reactive |
Non-reactive |
Evidence of immunity. No evidence of recent infection/recent vaccination |
|
Non-reactive |
Reactive |
Evidence of recent infection or vaccination. |
|
Reactive |
Reactive |
Evidence of recent infection or vaccination |
A positive result for HAV IgM with or without a positive HAV IgG suggests recent/acute infection with HAV. It may also be due to non-specificity of the test assay as a result of non-specific cross-reacting antibodies (e.g. rheumatoid factor, polyclonal antibody, etc.). A positive HAV IgM must be interpreted with clinical symptoms and/or exposure history.
Reporting
Results are reported to the physician, authorized health care provider (General O. Reg 45/22, s.18) or submitter as indicated on the requisition.
Specimens that are reactive for HAV IgM are reported to the Medical Officer of Health as per the Ontario Health Protection and Promotion Act.
Due to changes in both PHO’s and Ontario’s laboratory information systems, cumulative reporting is no longer available.
Vous n'avez pas de compte MonSPO? S'inscrire maintenant